Page 147 - JSOM Winter 2018
P. 147
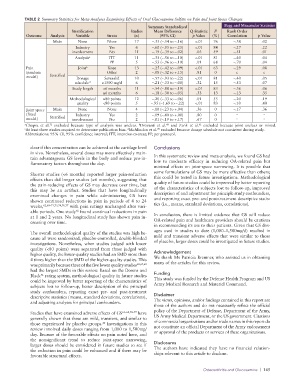
TABLE 2 Summary Statistics for Meta-Analyses Examining Effects of Oral Glucosamine Sulfate on Pain and Joint Space Changes
Summary Standardized Begg and Mazumdar Statistics
Stratification Studies Mean Difference Q-Statistic I 2 Rank Order
Outcome Analysis Variable Strata (n) (95% CI) p Value (%) Correlation p Value
Main None None 17 –.35 (–.54 to –.16) <.01 76 –.38 .02
Industry Yes 6 –.60 (–.95 to –.25) <.01 84 –.27 .22
involvement No 11 –.19 (–.39 to –.02) .03 59 –.51 .01
Analysis a ITT 11 –.33 (–.56 to –.10) <.01 81 –.40 .04
PP 5 –.53 (–.96 to –.10) .01 68 –.70 .04
Pain Joint b Knee 13 –.25 (–.42 to –.09) <.01 63 –.29 .08
(random Other 2 –.08 (–.32 to +.15) .81 0 c c
model) Stratified Dosage Several/d 10 –.57 (–.93 to –.22) <.01 81 –.40 .05
schedule d ≥1500 mg/d 6 –.21 (–.33 to –.08) .32 15 –.53 .07
Study length <6 months 11 –.54 (–.88 to –.19) <.01 83 –.36 .06
≥6 months 6 –.18 (–.30 to –.05) .33 13 –.13 .35
Methodological ≥80 points 12 –.20 (–.33 to –.06) .01 55 –.20 .19
quality <80 points 5 –.95 (–1.69 to –.22) <.01 83 –.10 .40
Joint space Main None None 4 –.10 (–.23 to +.04) .56 0 –.17 .36
(fixed Industry Yes 2 –.19 (–.40 to +.00) .80 0 c c
model) Stratified involvement No 2 –.01 (–.19 to +.17) .67 0 c c
62
72
b
68
a Chopra et al. excluded because type of analysis was unclear. Drovanti et al. and Zenk et al. excluded because joint unclear or mixed.
c At least three studies required to determine publication bias. McAlindon et al. excluded because dosage schedule not consistent during study.
d
76
Abbreviations: 95% CI, 95% confidence interval; ITT, intention-to-treat; PP, per protocol.
clear if this concentration can be achieved at the cartilage level Conclusions
in vivo. Nonetheless, several doses may more effectively main-
tain advantageous GS levels in the body and reduce pro-in- In this systematic review and meta-analysis, we found GS had
flammatory factors throughout the day. low to moderate efficacy in reducing OA-related pain but
minimal effects on joint-space narrowing. It is possible that
Shorter studies (<6 months) reported larger pain-reduction some formulations of GS may be more effective than others;
effects than did longer studies (≥6 months), suggesting that this could be tested in future investigations. Methodological
the pain-reducing effects of GS may decrease over time, but quality of future studies could be improved by better reporting
this may be an artifact. Studies that have longitudinally of the characteristics of subjects lost to follow-up, improved
examined changes in pain while administering GS have description of and adjustment for principle study confounders,
shown continued reductions in pain in periods of 4 to 24 and reporting exact pre- and post-treatment descriptive statis-
weeks, 62,68–71,73,74,76,77 with pain ratings unchanged after vari- tics (i.e., means, standard deviations, correlations).
able periods. One study found continual reductions in pain
66
at 1 and 2 years. No longitudinal study has shown pain in- In conclusion, there is limited evidence that GS will reduce
creasing over time. OA-related pain and healthcare providers should be cautious
in recommending its use to their patients. Given that GS dos-
The overall methodological quality of the studies was high be- ages used in studies to date (1,000–1,500mg/d) resulted in
cause all were randomized, placebo-controlled, double-blinded mild and transient adverse effects that were similar to those
investigations. Nonetheless, when studies judged with lower of placebo, larger doses could be investigated in future studies.
quality (<80 points) were separated from those judged with
higher quality, the lower-quality studies had an SMD more than Acknowledgement
4 times higher than the SMD of the higher-quality studies. This We thank Ms Patricia Bremner, who assisted us in obtaining
was primarily because three of the five lower quality studies 62,67,68 many of the articles for this review.
had the largest SMDs in this review. Based on the Downs and
Black rating system, methodological quality in future studies Funding
52
could be improved by better reporting of the characteristics of This study was funded by the Defense Health Program and US
subjects lost to follow-up, better description of the principal Army Medical Research and Materiel Command.
study confounders, reporting exact pre- and post-treatment
descriptive statistics (means, standard deviations, correlations), Disclaimer
and adjusting analyses for principal confounders. The views, opinions, and/or findings contained in this report are
those of the authors and do not necessarily reflect the official
Studies that have examined adverse effects of GS 63–68,70–77 have policy of the Department of Defense, Department of the Army,
generally shown that these are mild, transient, and similar to US Army Medical Department, or the US government. Citations
those experienced by placebo groups. Investigations in this of commercial organizations and/or trade names in this report do
48
review involved daily doses ranging from 1,000 to 1,500mg/ not constitute an official Department of the Army endorsement
day. Because of the favorable effects on pain noted here, and or approval of the products or services of these organizations.
the nonsignificant trend to reduce joint-space narrowing,
larger doses should be considered in future studies to see if Disclosures
the reduction in pain could be enhanced and if there may be The authors have indicated they have no financial relation-
favorable structural effects. ships relevant to this article to disclose.
Osteoarthritis and Glucosamine | 145