Page 145 - JSOM Winter 2018
P. 145
Quality Score (% of total points) b 55 69 85 80 88 87 90 76 85 87 82 74 90 83 78 83 86
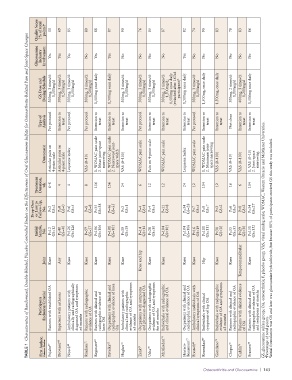
TABLE 1 Characteristics of Randomized, Double-Blinded, Placebo-Controlled Studies on the Effectiveness of Oral Glucosamine Sulfate for Osteoarthritis-Related Pain and Joint-Space Changes
Glucosamine Industry Involvement Yes Yes Yes No Yes Yes No No No No Yes No No No No No No
GS Dose and Dosing Schedule 500mg, 3 times/d: 1,500mg/d 500mg, 3 times/d: 1,500mg/d 500mg, 3 times/d: 1,500mg/d 500mg, 3 times/d: 1,500mg/d 1,500mg once daily 1,500mg once daily 500mg, 3 times/d: 1,500mg/d 500mg, 3 times/d: 1,500mg/d 500mg, 3 times/d: 1,500mg/d 500mg, 3 times/d: 1,500mg/d; 1,500mg once daily (switched after 163rd participant) b 1,500mg once daily 500mg, 3 times/d: 1,500mg/d 1,500mg, once daily
Type of Analysis Per protocol Intention to treat Per protocol Per protocol Intention to treat Intention to treat Intention to treat Per protocol Intention to treat Intention to treat Intention to treat Per protocol Intention to treat Intention to treat Not clear Intention to treat Intention to treat
Outcome(s) Articular pain on 4-point scale Articular pain on 4-point scale Lequesne index VAS (0–10) WOMAC pain scale Mean joint-space narrowing WOMAC pain scale Narrowest joint- space width VAS (0–100) WOMAC pain scale Pain on 4-point scale WOMAC pain scale Lequesne Index WOMAC pain scale WOMAC pain scale Minimum joint- space narrowing VAS (0–100) VAS (0–10) VAS (0–100) 1. VAS (0–10) Joint-space narrowing
Treatment Duration, wk 6–8 4 4 8 1. 156 2. 1. 156 2. 24 6 12 12 24 12 1. 104 2. 12 16 6 104 2.
Drop-Outs or Lost to Follow-Up, No. P=2 GS=2 P=0 GS=0 P=6 GS=5 P=8 GS=8 P=35 GS=38 P=46 GS=35 P=2 GS=1 P=4 GS=1 P=4 GS=3 P=11 GS=8 P=34 GS=28 P=7 GS=5 P=8 GS=7 P=2 GS=2 P=6 GS=9 P=8 GS=2 P=34 GS=27
Initial Sample, No. P=12 GS=12 P=40 GS=40 P=126 GS=126 P=57 GS=57 P=106 GS=106 P=101 GS=101 P=39 GS=39 P=14 GS=14 P=28 GS=30 P=104 GS=101 P=104 GS=106 P=16 GS=19 P=111 GS=111 P=30 GS=30 P=35 GS=35 P=29 GS=30 P=151 GS=152 GS, glucosamine sulfate group; OA, osteoarthritis; P, placebo group; VAS, visual analog scale; WOMAC, Western Ontario and McMaster Universities.
Knee and hip
Joint Knee Any Knee Knee Knee Knee Knee Knee Knee Knee Knee Hip Knee Knee Temporomandibular Knee b Initial formulation was GS and later was glucosamine hydrochloride, but because 80% of participants received GS this study was included.
Participants (Inclusion Criteria) Patients with established OA Inpatients with arthrosis Ambulatory patients with clinically and radiographically diagnosed OA and symptoms ≥6 months Patients with radiographic evidence of OA Patients with clinical and radiographic evidence of knee OA Outpatients with clinical and radiographic evidence of knee Ambulatory patients with clinical and radiographic evidence of OA and symptoms ≥3 months Out
First Author, Reference Number Pujalte 67 Drovanti 68 Noack 69 Rindone 71 Reginster 64 Pavelka 65 OA Hughes 73 Zenk 62 Usha 70 McAlindon 76 Herrero- Beaumont 77 Frestedt 63 Rozendaal 60 Giordano 74 Chopra 72 Cahlin 75 Fransen 66 a Percentage of total points.
Osteoarthritis and Glucosamine | 143