Page 62 - JSOM Spring 2020
P. 62
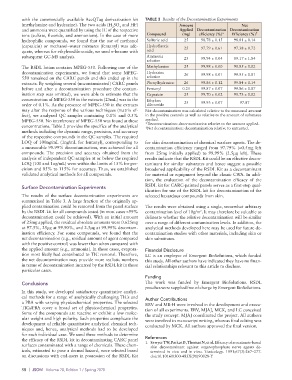
with the commercially available AccQTag derivatization kit TABLE 3 Results of the Decontamination Experiments
(methylamine and hydrazine). The two acids (H SO and HF) Amount Net
4
2
and ammonia were quantified by using the IC of the respective Applied Decontamination Decontamination
ions (sulfate, fluoride, and ammonium). In the case of more Compound (mg) efficiency (%)* Efficiency (%) †
hydrophilic compounds, we found that the use of methanol Sulfuric acid 25 98.78 ± 0.15 98.81 ± 0.14
(capsaicin) or methanol–water mixtures (fentanyl) was ade- Hydrofluoric 25 97.79 ± 0.61 97.38 ± 0.72
quate, whereas for ethylenedibromide, we used n-hexane with acid
subsequent GC-MS analysis. Ammonia 25 99.94 ± 0.04 89.17 ± 1.84
solution
The RSDL lotion contains MPEG-550. Following one of the Methylamine 25 99.98 ± 0.00 99.85 ± 0.02
decontamination experiments, we found that some MPEG- Hydrazine 26 99.98 ± 0.01 99.95 ± 0.01
550 remained on the CARC panels and this ended up in the solution
extracts. By weighing several (uncontaminated) CARC panels Phenylhydrazine 26 99.86 ± 0.12 99.84 ± 0.14
before and after a decontamination procedure (the contam- Fentanyl 0.25 99.87 ± 0.07 99.86 ± 0.07
ination step was omitted), we were able to estimate that the Capsaicin 25 99.70 ± 0.02 99.73 ± 0.02
concentration of MPEG-550 in the extracts (25mL) was in the Ethylene
order of 0.1%. As the presence of MPEG-550 in the extracts dibromide 25 99.93 ± 0.07 97.87
may alter the response of the various techniques (matrix ef- Net decontamination was calculated relative to the measured amount
fect), we analyzed QC samples containing 0.1% and 0.5% in the positive controls as well as relative to the amount of substance
MPEG-550. No interference of MPEG-550 was found at these applied.
concentrations. Table 2 provides the specifics of the analytical *Decontamination: decontamination relative to the amount applied.
†
Net decontamination: decontamination relative to untreated.
methods including the dynamic range, precision, and accuracy
of the respective compounds in the QC samples. The required
LOQ of 100ng/mL (1ng/mL for fentanyl), corresponding to for skin decontamination of chemical warfare agents. The de-
a measurable 99.99% decontamination, was achieved for all contamination efficiency ranged from 97.79% (≈0.5mg left
compounds. The precision and accuracy obtained from the from 25mg initially applied) to 99.99% (2.5µg left). These
analysis of independent QC samples at or below the required results indicate that the RSDL Kit could be an effective decon-
LOQ (100 and 1ng/mL) were within the limits of 15% for pre- taminant for similar substrates and hence suggest a possible
cision and 85% to 115% for accuracy. Thus, we established broadened applicability of the RSDL Kit as a decontaminant
validated analytical methods for all compounds. for material or equipment beyond the classic CWA. In addi-
tion, the evaluation of the decontamination efficacy of the
Surface Decontamination Experiments RSDL kit for CARC-painted panels serves as a first-step qual-
ification for use of the RSDL kit for decontamination of the
The results of the surface decontamination experiments are selected hazardous compounds from skin.
summarized in Table 3. A large fraction of the originally ap-
plied contamination could be removed from the panel surface The results were obtained using a single, somewhat arbitrary
by the RSDL kit for all compounds tested (in most cases >99% contamination level of 10g/m . It may therefore be valuable to
2
decontamination could be achieved). With an initial amount delineate whether the relative decontamination will be similar
of 25mg applied, the residual absolute amounts were 0.625mg over a range of different contamination levels. In addition, the
at 97.5%, 25µg at 99.90%, and 2.5µg at 99.99% decontam- analytical methods developed here may be used for future de-
ination efficiency. For some compounds, we found that the contamination studies with other materials, including skin or
net decontamination (e.g., residual amount of agent compared skin substitutes.
with the positive control) was lower than when compared with
the applied amount (e.g., ammonia). In those cases, evapora- Financial Disclosure
tion most likely had contributed to TIC removal. Therefore, LC is an employee of Emergent BioSolutions, which funded
the net decontamination may provide more realistic numbers this study. All other authors have indicated they have no finan-
in terms of decontamination incurred by the RSDL kit in those cial relationships relevant to this article to disclose.
particular cases.
Funding
Conclusions The work was funded by Emergent BioSolutions. RSDL
pouches were supplied free of charge by Emergent BioSolutions.
In this study, we developed satisfactory quantitative analyti-
cal methods for a range of analytically challenging TICs and Author Contributions
a PBA with varying physicochemical properties. The selected ERV and MB-H were involved in the development and execu-
TICs/PBA cover a broad set of physicochemical properties. tion of all experiments. ERV, MJAJ, MCK, and LC conceived
Some of the compounds are reactive or exhibit a low molec- the study concept. MJAJ coordinated the project. All authors
ular weight and high polarity. Such properties complicate the were involved in manuscript writing, whereas final editing was
development of reliable quantitative analytical chemical tech- conducted by MCK. All authors approved the final version.
niques and, hence, analytical methods had to be developed
for each individual case. We used these methods to determine
the efficacy of the RSDL kit in decontaminating CARC panel References
surfaces contaminated with a range of chemicals. These chem- 1. Sawyer TW, Parker D, Thomas N, et al. Efficacy of an oximate-based
skin decontaminant against organophosphate nerve agents de-
icals, estimated to pose a dermal hazard, were selected based termined in vivo and in vitro. Toxicology. 1991;67(3):267–277.
on discussions with end-users in possession of the RSDL Kit doi:10.1016/0300-483X(91)90028-Y
58 | JSOM Volume 20, Edition 1 / Spring 2020