Page 141 - JSOM Spring 2020
P. 141
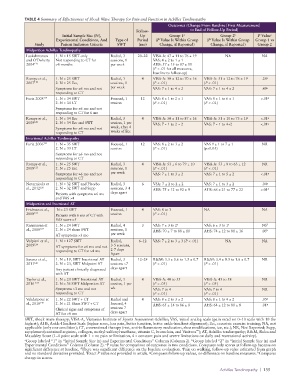
TABLE 4 Summary of Effectiveness of Shock Wave Therapy for Pain and Function in Achilles Tendinopathy
Outcomes (Change From Baseline [First Measurement]
Follow- to End of Follow-Up Period)
Initial Sample Size (N), Up Group 1 Group 2 b P Value c
a
Experimental Conditions, And Type of Period (P Value Is Within Group (P Value Is Within Group Group 1 vs
Study Patient Inclusion Criteria SWT (mo) Change, if Reported) Change, if Reported) Group 2
Midportion Achilles Tendinopathy
Laskshmanan 1. N = 15 SWT only Radial, 3 20–22 VISA-A: 47 ± 11 to 76 ± 19 NA NA
and O’Doherty Not responding to CT for sessions, 1 VAS: 4 ± 2 to 1 ± 1
2004 117 >6 months per week AHS: 57 ± 15 to 87 ± 11
(P < .01 for all measures,
baseline to follow-up)
Rompe et al., 1. N = 25 SWT Radial, 3 4 VISA-A: 50 ± 12 to 70 ± 16 VISA-A: 51 ± 12 to 76 ± 19 .26 g
2007 118 2. N = 25 Ecc, sessions, 1 (P < .01) (P < .01)
Symptoms for ≥6 mo and not per week VAS: 7 ± 1 to 4 ± 2 VAS: 7 ± 1 to 4 ± 2 .49 g
responding to CT
Furia 2008 119 1. N = 34 SWT Focused, 1 12 VAS: 8 ± 1 to 2 ± 1 VAS: 8 ± 1 to 6 ± 1 <.01 g
2. N = 34 CT session (P < .01) (P < .01)
Symptoms for ≥6 mo and not
responding to CT for 6 mo
Rompe et al., 1. N = 34 Ecc Radial, 3 4 VISA-A: 50 ± 11 to 87 ± 16 VISA-A: 51 ± 10 to 73 ± 19 <.01 g
2009 120 2. N = 34 Ecc and SWT session, 1 per VAS: 7 ± 1 to 2 ± 2 VAS: 7 ± 1 to 4+2 <.01 g
Symptoms for ≥6 mo and not week, after 4
responding to CT weeks of Ecc
Insertional Achilles Tendinopathy
Furia 2006 121 1. N = 35 SWT Focused, 1 12 VAS: 8 ± 2 to 3 ± 2 VAS 9 ± 1 to 7 ± 1 NR
2. N = 33 CT session (P < .01) (p>0.05)
Symptoms for ≥6 mo and not
responding to CT
Rompe et al., 1. N = 25 SWT Radial, 3 4 VISA-A: 53 ± 6 to 79 ± 10 VISA-A: 53 ± 8 to 63 ± 12 NR
2008 122 2. N = 25 Ecc sessions, 1 (P < .01) (P < .01)
Symptoms for ≥6 mo and not per week VAS: 7 ± 1 to 3 ± 2 VAS: 7 ± 1 to 5 ± 2 <.01 g
responding to CT
Notarnicola et 1. N = 32 SWT and Placebo Radial, 3 6 VSA: 7 ± 2 to 2 ± 2 VAS: 7 ± 1 to 3 ± 2 .04 g
al., 2012 126 2. N = 32 SWT and Supp sessions, 3-4 AHS: 75 ± 12 to 92 ± 9 AHS: 66 ± 21 to 77 ± 22 <.01 d
Patients with symptoms ≥6 mo days apart
and VAS >4
Midportion and Insertional AT
Fridman et al., N = 23 SWT Focused, 1 4 VAS: 8 to 3 NA NA
2008 123 Patients with 6 mo of CT with session (P < .01)
VAS score >5
Rasmussen et 1. N = 24 SWT Radial, 4 3 VAS: 7 ± 3 to 2 e VAS: 6 ± 3 to 1 e NS f
al., 2008 127 2. N = 24 sham SWT sessions, 1 AHS: 70 ± 7 to 88 ± 10 AHS: 74 ± 12 to 81 ± 16 .05 h
AT symptoms >3 mo per week
Vulpiani et al., 1. N = 127 SWT Radial, 6–12 VAS: 7 ± 2 to 3 ± 3 (P < .01) NA NA
2009 124 AT symptoms for ≥6 mo and not 3-5 sessions,
responding to CT for >4 mo 2-7 days
Apart
Saxena et al., 1. N = 19, SWT Insertional AT Radial, 3 12–24 R&M: 3.3 ± 0.6 to 1.5 ± 0.7 R&M: 3.4 ± 0.5 to 1.6 ± 0.7 NR
2011 128 2. N = 23, SWT Midpoint AT sessions ~7 (P < .01) (P < .01)
Any patient clinically diagnosed days apart
with AT
Taylor et al., 1. N = 20 SWT Insertional AT Radial, 3 4 VISA-A: 40 to 55 VISA-A: 43 to 58 NR
2016 125 2. N = 36 SWT Midportion AT session, 1 per (P < .05) (P < .05)
Symptoms >3 mo and not wk VAS: 7 to 4 VAS: 7 to 4 NR
responding to CT (P < .01) (P < .01)
Vahdatpour et 1. N = 22 SWT + CT Radial and 4 VAS: 8 ± 2 to 3 ± 2 VAS: 8 ± 1 to 4 ± 2 .05 g
al., 2018 149 2. N = 21 Sham SWT + CT focused, 4 AHS: 65 ± 14 to 86 ± 8 AHS: 64 ± 12 to 80 ± 8 .01 g
Clinical signs and symptoms of sessions 7
AT for >6 mo days apart
SWT, shock wave therapy; VISA-A, Victorian Institute of Sports Assessment-Achilles; VAS, visual analog scale (pain rated on 0–10 scale with 10 the
highest); AHS, Ankle-Hindfoot Scale (higher score, less pain, better function, better ankle-hindfoot alignment); Ecc, eccentric exercise training; NA, not
applicable (only one condition); CT, conventional therapy (rest, anti-inflammatory medication, shoe modifications, ice, etc.); NR, Not Reported; Supp,
supplement(contained arginine, collagen, methyl-sulfonyl-methane, vitamin C, bromelain, and Vinitrox ); AT, Achilles tendinopathy; R&M, Roles and
™
Maudsley Score (1–4 point scale with 1 = no pain or limitation, 4 = constant pain and severe limitations on daily and recreational activity).
a Group labeled “1” in “Initial Sample Size (n) and Experimental Conditions” Column (Column 2). Group labeled “2” in “Initial Sample Size (n) and
b
Experimental Conditions” Column (Column 2). P value for comparison of outcomes in two conditions. Compares only scores at follow-up because no
c
significant difference at baseline. There was significant difference on the baseline evaluation. Pain on walking, follow-up value estimated from graph
e
d
h
and no standard deviation provided. Exact P value not provided in article. Compares follow-up values, no difference on baseline measures. Compares
g
f
change in scores.
Achilles Tendinopathy | 135