Page 56 - Journal of Special Operations Medicine - Summer 2016
P. 56
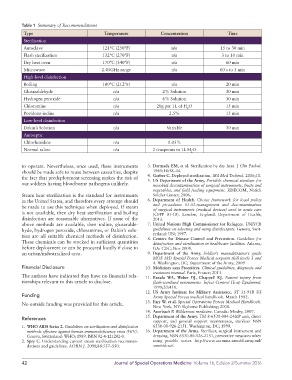
Table 1 Summary of Recommendations
Type Temperature Concentration Time
Sterilization
Autoclave 121°C (250°F) n/a 15 to 30 min
Flash sterilization 132°C (270°F) n/a 3 to 10 min
Dry heat oven 170°C (340°F) n/a 60 min
Microwave 2.45GHz range n/a 60 s to 5 min
High-level disinfection
Boiling 100°C (212°F) n/a 20 min
Glutaraldehyde n/a 2% Solution 30 min
Hydrogen peroxide n/a 6% Solution 30 min
Chloramine n/a 20g per 1L of H O 15 min
2
Povidone-iodine n/a 2.5% 15 min
Low-level disinfection
Dakin’s Solution n/a Variable 30 min
Antiseptic
Chlorhexidine n/a 0.05%
Normal saline n/a 2 teaspoons in 1L H O
2
to operate. Nevertheless, once used, these instruments 3. Darmady EM, et al. Sterilization by dry heat. J Clin Pathol.
should be made safe to reuse between casualties, despite 1961;14:38–44.
the fact that predeployment screening makes the risk of 4. Gerber C. Deployed sterilization. Mil Med Technol. 2006;10.
5. US Department of the Army, Portable chemical sterilizer for
our soldiers having bloodborne pathogens unlikely. micobial decontamination of surgical instruments, fruits and
vegetables, and field feeding equipment. RDECOM, Natick
Steam heat sterilization is the standard for instruments Soldier Center; 2006.
in the United States, and therefore every attempt should 6. Department of Health. Choice framework for local policy
be made to use this technique when deployed. If steam and procedures 01-01-management and decontamination
of surgical instruments (medical devices) used in acute care
is not available, then dry heat sterilization and boiling (CFPP 01-10). London, England: Department of Health;
disinfection are reasonable alternatives. If none of the 2013.
above methods are available, then iodine, glutaralde- 7. United Nations High Commissioner for Refugees. UNHCR
hyde, hydrogen peroxide, chloramines, or Dakin’s solu- guidelines on selecting and using disinfectants. Geneva, Swit-
zerland: UN; 1997.
tion are all suitable chemical methods of disinfection. 8. Centers for Disease Control and Prevention. Guideline for
These chemicals can be stocked in sufficient quantities disinfection and sterilization in healthcare facilities. Atlanta,
before deployment or can be procured locally if close to GA: CDC; Nov 2008.
an urban/industrialized area. 9. Department of the Army. Soldier’s manual/trainer’s guide
MOS 18D Special Forces Medical sergeant skill levels 3 and
4. Washington, DC; Department of the Army; 2009
Financial Disclosure 10. Médicines sans Frontières. Clinical guidelines, diagnosis and
treatment manual. Paris, France; 2013.
The authors have indicated they have no financial rela- 11. Rutala WA, Weber DJ, Chappell KJ. Patient injury from
tionships relevant to this article to disclose. flash-sterilized instruments. Infect Control Hosp Epidemiol.
1999;20:458.
12. US Army Institute for Military Assistance. ST 31-91B US
Funding Army Special Forces medical handbook. March 1982.
No outside funding was provided for this article. 13. Farr W, et al. Special Operations Forces Medical Handbook.
New York, NY: Skyhorse Publishing; 2008.
14. Auerbach P. Wilderness medicine. Canada: Mosby; 2007.
References 15. Department of the Army. TM 8-6530-004-24&P unit, direct
support, and general support maintenance, sterilizer NSN
1. WHO AIDS Series 2. Guidelines on sterilization and disinfection 6530-00-926-2151. Washington, DC; 1990.
methods effective against human immunodeficiency virus (HIV). 16. Department of the Army. Sterilizer, surgical instrument and
Geneva, Switzerland: WHO; 1989. ISBN 92-4-121202-0. dressing, NSN 6530-00-926-2151, preventive measures when
2. Spry C. Understanding current steam sterilization recommen- using potable water. http://www.usamma.amedd.army.mil/
dations and guidelines. AORN J. 2008;88:537–550. assets/docs/.
42 Journal of Special Operations Medicine Volume 16, Edition 2/Summer 2016