Page 40 - JSOM Spring 2021
P. 40
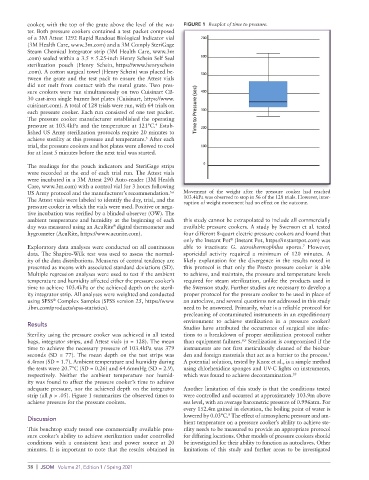
cooker, with the top of the grate above the level of the wa- FIGURE 1 Boxplot of time to pressure.
ter. Both pressure cookers contained a test packet composed
of a 3M Attest 1292 Rapid Readout Biological Indicator vial
(3M Health Care, www.3m.com) and a 3M Comply SteriGage
Steam Chemical Integrator strip (3M Health Care, www.3m
.com) sealed within a 3.5 ´ 5.25-inch Henry Schein Self Seal
sterilization pouch (Henry Schein, https://www.henryschein
.com). A cotton surgical towel (Henry Schein) was placed be-
tween the grate and the test pack to ensure the Attest vials
did not melt from contact with the metal grate. Two pres-
sure cookers were run simultaneously on two Cuisinart CB-
30 cast-iron single burner hot plates (Cuisinart, https://www.
cuisinart.com). A total of 128 trials were run, with 64 trials on
each pressure cooker. Each run consisted of one test packet.
The pressure cooker manufacturer established the operating
pressure at 103.4kPa and the temperature at 121°C. Estab-
4
lished US Army sterilization protocols require 20 minutes to
achieve sterility at this pressure and temperature. After each
5
trial, the pressure cookers and hot plates were allowed to cool
for at least 5 minutes before the next trial was started.
The readings for the pouch indicators and SteriGage strips
were recorded at the end of each trial run. The Attest vials
were incubated in a 3M Attest 290 Auto-reader (3M Health
Care, www.3m.com) with a control vial for 3 hours following
US Army protocol and the manufacturer’s recommendation. Movement of the weight after the pressure cooker had reached
5,6
The Attest vials were labeled to identify the day, trial, and the 103.4kPa was observed to stop in 56 of the 128 trials. However, inter-
pressure cooker in which the vials were used. Positive or nega- ruption of weight movement had no effect on the outcome.
tive incubation was verified by a blinded observer (OW). The
ambient temperature and humidity at the beginning of each this study cannot be extrapolated to include all commercially
day was measured using an AcuRite digital thermometer and available pressure cookers. A study by Swenson et al. tested
®
hygrometer (AcuRite, https://www.acurite.com). four different 8-quart electric pressure cookers and found that
only the Instant Pot (Instant Pot, https://instantpot.com) was
®
Exploratory data analyses were conducted on all continuous able to inactivate G. sterothermophilus spores. However,
7
data. The Shapiro-Wilk test was used to assess the normal- sporicidal activity required a minimum of 120 minutes. A
ity of the data distributions. Measures of central tendency are likely explanation for the divergence in the results noted in
presented as means with associated standard deviations (SD). this protocol is that only the Presto pressure cooker is able
Multiple regression analyses were used to test if the ambient to achieve, and maintain, the pressure and temperature levels
temperature and humidity affected either the pressure cooker’s required for steam sterilization, unlike the products used in
time to achieve 103.4kPa or the achieved depth on the steril- the Swenson study. Further studies are necessary to develop a
ity integrator strip. All analyses were weighted and conducted proper protocol for the pressure cooker to be used in place of
using SPSS Complex Samples (SPSS version 25, https://www an autoclave, and several questions not addressed in this study
®
.ibm.com/products/spss-statistics). need to be answered. Primarily, what is a reliable protocol for
precleaning of contaminated instruments in an expeditionary
environment to achieve sterilization in a pressure cooker?
Results
Studies have attributed the occurrence of surgical site infec-
Sterility using the pressure cooker was achieved in all tested tions to a breakdown of proper sterilization protocol rather
bags, integrator strips, and Attest vials (n = 128). The mean than equipment failures. Sterilization is compromised if the
8,9
time to achieve the necessary pressure of 103.4kPa was 379 instruments are not first meticulously cleaned of the biobur-
seconds (SD = 77). The mean depth on the test strips was den and foreign materials that act as a barrier to the process.
1
6.4mm (SD = 1.7). Ambient temperature and humidity during A potential solution, tested by Knox et al., is a simple method
the tests were 20.7°C (SD = 0.26) and 64.6mmHg (SD = 2.9), using chlorhexidine sponges and UV-C lights on instruments,
respectively. Neither the ambient temperature nor humid- which was found to achieve decontamination. 10
ity was found to affect the pressure cooker’s time to achieve
adequate pressure, nor the achieved depth on the integrator Another limitation of this study is that the conditions tested
strip (all p > .05). Figure 1 summarizes the observed times to were controlled and occurred at approximately 103.9m above
achieve pressure for the pressure cookers. sea level, with an average barometric pressure of 0.996atm. For
every 152.4m gained in elevation, the boiling point of water is
lowered by 0.03°C. The effect of atmospheric pressure and am-
8
Discussion
bient temperature on a pressure cooker’s ability to achieve ste-
This benchtop study tested one commercially available pres- rility needs to be measured to provide an appropriate protocol
sure cooker’s ability to achieve sterilization under controlled for differing locations. Other models of pressure cookers should
conditions with a consistent heat and power source at 20 be investigated for their ability to function as autoclaves. Other
minutes. It is important to note that the results obtained in limitations of this study and further areas to be investigated
38 | JSOM Volume 21, Edition 1 / Spring 2021