Page 23 - Journal of Special Operations Medicine - Winter 2015
P. 23
according to their clinical judgment, current FDA reg- 3. Efforts to leverage technology and to develop elec-
ulations prohibit medications from being marketed or tronic methods of capturing prehospital medical care
packaged by their manufacturer for off-label uses. This should be encouraged and funded.
is an very significant problem on the battlefield, where
combat medics, corpsmen, and PJs provide the vast Reliable documentation of care rendered in the prehos-
majority of prehospital combat casualty care under the pital environment is critical but has proven difficult to
most challenging circumstances imaginable. Medica- accomplish. An accurate record of prehospital care ren-
tions being used for off-label indications, such as sub- dered is important for several reasons: (1) it may help
dissociative doses of ketamine for analgesia, cannot be guide further care that will be rendered to the casualty
packaged in autoinjectors or other formats that facili- at medical treatment facilities; (2) prehospital care is an
tate their use for off-label uses. This regulatory anomaly essential part of the casualty’s electronic health record;
therefore requires Combat medical providers to draw and (3) accurate records of prehospital care are crucial
up doses of medications on the battlefield from mul- to combat casualty care performance improvement ef-
tidose containers in the middle of battlefield casualty forts conducted by the military’s JTS.
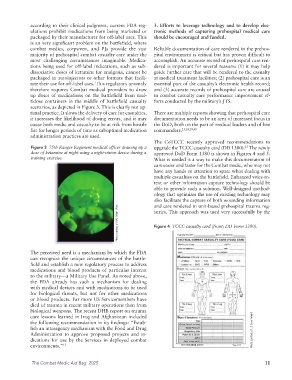
scenarios, as depicted in Figure 3. This is clearly not op-
timal practice. It slows the delivery of care for casualties, There are multiple reports showing that prehospital care
it increases the likelihood of dosing errors, and it may documentation needs to be an area of increased focus in
cause both medic and casualty to be at risk from hostile the DoD, both on the part of medical leaders and of line
fire for longer periods of time as suboptimal medication commanders. 25,28,29,40
administration practices are used.
The CoTCCC recently approved recommendations to
Figure 3 75th Ranger Regiment medical officer drawing up a upgrade the TCCC casualty card (DD 1380). The newly
25
dose of ketamine at night using a night-vision device during a approved DoD Form 1380 is shown in Figures 4 and 5.
training exercise. What is needed is a way to make this documentation of
care easier and faster for the Combat medic, who may not
have any hands or attention to spare when dealing with
multiple casualties on the battlefield. Enhanced voice-to-
text or other information capture technology should be
able to provide such a solution. Well-designed method-
ology that optimizes the use of existing technology may
also facilitate the capture of both wounding information
and care rendered in unit-based prehospital trauma reg-
Photo courtesy of MAJ Ethan Miles Figure 4 TCCC casualty card (front; DD Form 1380).
istries. This approach was used very successfully by the
The perceived need is a mechanism by which the FDA
can recognize the unique circumstances of the battle-
field and establish a new regulatory process to address
medications and blood products of particular interest
to the military—a Military Use Panel. As noted above,
the FDA already has such a mechanism for dealing
with medical devices and with medications to be used
for biological threats, but not for other medications
or blood products. Far more US Servicemembers have
died of trauma in recent military operations than from
biological weapons. The recent DHB report on trauma
care lessons learned in Iraq and Afghanistan included
the following recommendation in its findings: “Estab-
lish an interagency mechanism with the Food and Drug Photo courtesy of US Government
Administration to approve proposed projects and in-
dications for use by the Services in deployed combat
environments.” 15
The Combat Medic Aid Bag: 2025 11