Page 79 - JSOM Spring 2026
P. 79
That said, a study uncovered in our literature review by Lv
et al. described the use of a portable ultrasound, the Philips
CX50 (Philips Medical Systems, Andover, MA). Based on
17
their report, the Philips CX50 had a contrast software pack-
age. Philips no longer manufactures or sells this model,
17
which is among the reasons it was not included in our methods
testing. Beyond this one model, however, our literature search
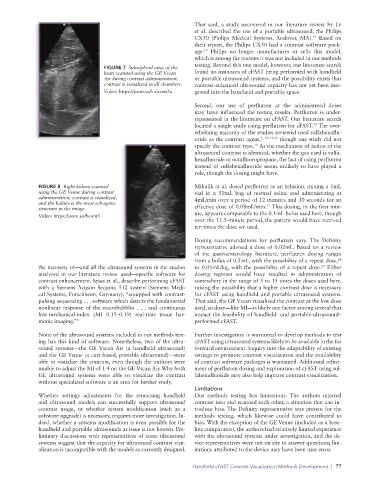
FIGURE 7 Subxiphoid view of the
heart scanned using the GE Vscan found no instances of cFAST being performed with handheld
Air during contrast administration; or portable ultrasound systems, and the possibility exists that
contrast is visualized in all chambers. contrast-enhanced ultrasound capacity has not yet been inte-
Video: https://jsom.us/kidcont2a grated into the handheld and portable space.
Second, our use of perflutren at the administered doses
may have influenced the testing results. Perflutren is under-
represented in the literature on cFAST. Our literature search
located a single study using perflutren for cFAST. The over-
11
whelming majority of the studies reviewed used sulfahexaflu-
oride as the contrast agent, 9, 12–17,23 though one study did not
specify the contrast type. As the mechanism of action of the
10
ultrasound contrast is identical, whether the gas used is sulfa-
hexafluoride or octafluoropropane, the fact of using perflutren
instead of sulfahexafluoride seems unlikely to have played a
role, though the dosing might have.
FIGURE 8 Right kidney scanned Mihalik et al. dosed perflutren as an infusion, mixing a 1mL
using the GE Venue during contrast vial in a 50mL bag of normal saline and administering at
administration; contrast is visualized, 4mL/min over a period of 12 minutes and 30 seconds for an
and the kidney is the most echogenic 11
structure in the image. effective dose of 0.08mL/min. This dosing, in the first min-
Video: https://jsom.us/hcont1 ute, appears comparable to the 0.1mL bolus used here, though
over the 12.5-minute period, the patient would have received
ten times the dose we used.
Dosing recommendations for perflutren vary. The Definity
representative advised a dose of 0.02mL. Based on a review
of the gastroenterology literature, perflutren dosing ranges
from a bolus of 0.5mL, with the possibility of a repeat dose,
26
the necessity of—and all the ultrasound systems in the studies to 0.01mL/kg, with the possibility of a repeat dose. Either
27
analyzed in our literature review used—specific software for dosing regimen would have resulted in administration of
contrast enhancement. Sessa et al., describe performing cFAST somewhere in the range of 5 to 15 times the doses used here,
with a Siemens Acuson Sequoia 512 system (Siemens Medi- raising the possibility that a higher contrast dose is necessary
cal Systems, Forschheim, Germany), “equipped with contrast- for cFAST using handheld and portable ultrasound systems.
pulsing sequencing . . . software which detects the fundamental That said, the GE Vscan visualized the contrast at the low dose
nonlinear response of the microbubbles . . . and continuous used, so dose—like MI—is likely one factor among several that
low-mechanical-index (MI 0.15–0.19) real-time tissue har- impact the feasibility of handheld- and portable- ultrasound–
monic imaging.” 9 performed cFAST.
None of the ultrasound systems included in our methods test- Further investigation is warranted to develop methods to test
ing has this kind of software. Nonetheless, two of the ultra- cFAST using ultrasound systems likely to be available in the far
sound systems—the GE Vscan Air (a handheld ultrasound) forward environment. Inquiry into the adaptability of existing
and the GE Venue (a cart-based, portable ultrasound)—were settings to promote contrast visualization and the availability
able to visualize the contrast, even though the authors were of contrast software packages is warranted. Additional refine-
unable to adjust the MI of 1.4 on the GE Vscan Air. Why both ment of perflutren dosing and exploration of cFAST using sul-
GE ultrasound systems were able to visualize the contrast fahexafluoride may also help improve contrast visualization.
without specialized software is an area for further study.
Limitations
Whether settings adjustments for the remaining handheld Our methods testing has limitations. The authors injected
and ultrasound models can successfully support ultrasound contrast into and scanned each other, a situation that can in-
contrast usage, or whether system modification (such as a troduce bias. The Definity representative was present for the
software upgrade) is necessary, requires more investigation. In- methods testing, which likewise could have contributed to
deed, whether a systems modification is even possible for the bias. With the exception of the GE Venue (included as a base-
handheld and portable ultrasounds at issue is not known. Pre- line comparator), the authors had relatively limited experience
liminary discussions with representatives of some ultrasound with the ultrasound systems under investigation, and the de-
systems suggest that the capacity for ultrasound contrast visu- vice representatives were not on site to answer questions; lim-
alization is incompatible with the models as currently designed. itations attributed to the device may have been user error.
Handheld cFAST Contrast Visualization Methods Development | 77