Page 77 - JSOM Spring 2026
P. 77
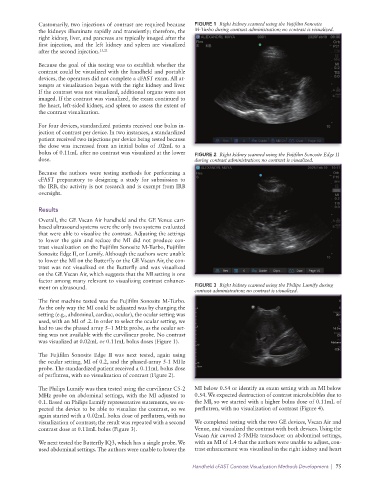
Customarily, two injections of contrast are required because FIGURE 1 Right kidney scanned using the Fujifilm Sonosite
the kidneys illuminate rapidly and transiently; therefore, the M-Turbo during contrast administration; no contrast is visualized.
right kidney, liver, and pancreas are typically imaged after the
first injection, and the left kidney and spleen are visualized
after the second injection. 15,21
Because the goal of this testing was to establish whether the
contrast could be visualized with the handheld and portable
devices, the operators did not complete a cFAST exam. All at-
tempts at visualization began with the right kidney and liver.
If the contrast was not visualized, additional organs were not
imaged. If the contrast was visualized, the exam continued to
the heart, left-sided kidney, and spleen to assess the extent of
the contrast visualization.
For four devices, standardized patients received one bolus in-
jection of contrast per device. In two instances, a standardized
patient received two injections per device being tested because
the dose was increased from an initial bolus of .02mL to a
bolus of 0.11mL after no contrast was visualized at the lower FIGURE 2 Right kidney scanned using the Fujifilm Sonosite Edge II
dose. during contrast administration; no contrast is visualized.
Because the authors were testing methods for performing a
cFAST preparatory to designing a study for submission to
the IRB, the activity is not research and is exempt from IRB
oversight.
Results
Overall, the GE Vscan Air handheld and the GE Venue cart-
based ultrasound systems were the only two systems evaluated
that were able to visualize the contrast. Adjusting the settings
to lower the gain and reduce the MI did not produce con-
trast visualization on the Fujifilm Sonosite M-Turbo, Fujifilm
Sonosite Edge II, or Lumify. Although the authors were unable
to lower the MI on the Butterfly or the GE Vscan Air, the con-
trast was not visualized on the Butterfly and was visualized
on the GE Vscan Air, which suggests that the MI setting is one
factor among many relevant to visualizing contrast enhance-
ment on ultrasound. FIGURE 3 Right kidney scanned using the Philips Lumify during
contrast administration; no contrast is visualized.
The first machine tested was the Fujifilm Sonosite M-Turbo.
As the only way the MI could be adjusted was by changing the
setting (e.g., abdominal, cardiac, ocular), the ocular setting was
used, with an MI of .2. In order to select the ocular setting, we
had to use the phased array 5–1 MHz probe, as the ocular set-
ting was not available with the curvilinear probe. No contrast
was visualized at 0.02mL or 0.11mL bolus doses (Figure 1).
The Fujifilm Sonosite Edge II was next tested, again using
the ocular setting, MI of 0.2, and the phased-array 5-1 MHz
probe. The standardized patient received a 0.11mL bolus dose
of perflutren, with no visualization of contrast (Figure 2).
The Philips Lumify was then tested using the curvilinear C5-2 MI below 0.54 or identify an exam setting with an MI below
MHz probe on abdominal settings, with the MI adjusted to 0.54. We expected destruction of contrast microbubbles due to
0.1. Based on Philips Lumify representative statements, we ex- the MI, so we started with a higher bolus dose of 0.11mL of
pected the device to be able to visualize the contrast, so we perflutren, with no visualization of contrast (Figure 4).
again started with a 0.02mL bolus dose of perflutren, with no
visualization of contrast; the result was repeated with a second We completed testing with the two GE devices, Vscan Air and
contrast dose at 0.11mL bolus (Figure 3). Venue, and visualized the contrast with both devices. Using the
Vscan Air curved 2-5MHz transducer on abdominal settings,
We next tested the Butterfly IQ3, which has a single probe. We with an MI of 1.4 that the authors were unable to adjust, con-
used abdominal settings. The authors were unable to lower the trast enhancement was visualized in the right kidney and heart
Handheld cFAST Contrast Visualization Methods Development | 75