Page 131 - JSOM Fall 2022
P. 131
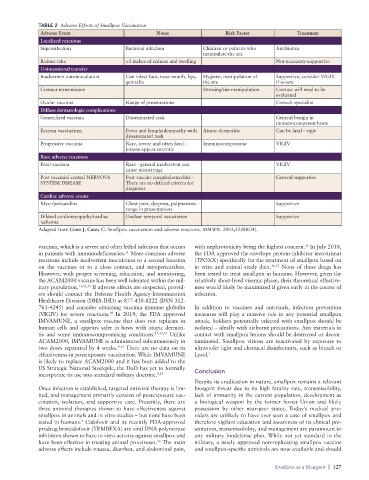
TABLE 2 Adverse Effects of Smallpox Vaccination
Adverse Event Notes Risk Factor Treatment
Localized reactions
Superinfection Bacterial infection Children or patients who Antibiotics
manipulate the site
Robust take >3 inches of redness and swelling Not necessarysupportive
Unintentional transfer
Inadvertent autoinoculation Can infect face, nose mouth, lips, Hygiene, manipulation of Supportive, consider VIGIV
genitalia the site if severe
Contact transmission Dressing/site manipulation Contact will need to be
evaluated
Ocular vaccinia Range of presentations Consult specialist
Diffuse dermatologic complications
Generalized vaccinia Disseminated rash General benign in
immunocompetent hosts
Eczema vaccinatum Fever and lymphadenopathy with Atopic dermatitis Can be fatal vigiv
disseminated rash
Progressive vaccinia Rare, severe and often fatal Immunocompromise VIGIV
lesions appear necrotic
Rare adverse reactions
Fetal vaccinia Rare general inadvertent can VIGIV
cause miscarriage
Post vaccinial central NERVOUS Post vaccine encephalomyelitis General supportive
SYSTEM DISEASE There are no defined criteria for
diagnosis
Cardiac adverse events
Myo/pericarditis Chest pain, dyspnea, palpitations Supportive
range in presentations
Dilated cardiomyopathy/cardiac Unclear temporal association Supportive
ischemia
Adapted from Cono J, Casey C. Smallpox vaccination and adverse reactions. MMWR. 2003;52(RR04).
vaccinia, which is a severe and often lethal infection that occurs with nephrotoxicity being the highest concern. In July 2018,
36
31
in patients with immunodeficiencies. More common adverse the FDA approved the envelope proteininhibitor tecovirimat
reactions include inadvertent inoculation to a second location (TPOXX) specifically for the treatment of smallpox based on
on the vaccinee or to a close contact, and myopericarditis. in vitro and animal study data. 36,37 None of these drugs has
However, with proper screening, education, and monitoring, been tested to treat smallpox in humans. However, given the
the ACAM2000 vaccine has been well tolerated within the mil relatively shortlived viremic phase, their theoretical effective
itary population. 13,32,33 If adverse effects are suspected, provid ness would likely be maximized if given early in the course of
ers should contact the Defense Health Agency Immunization infection.
Healthcare Division (DHAIHD) at 8774388222 (DSN 312
7614245) and consider obtaining vaccinia immune globulin In addition to vaccines and antivirals, infection prevention
(VIGIV) for severe reactions. In 2019, the FDA approved measures will play a massive role in any potential smallpox
30
IMVAMUNE, a smallpox vaccine that does not replicate in attack. Soldiers potentially infected with smallpox should be
human cells and appears safer in hosts with atopic dermati isolated – ideally with airborne precautions. Any materials in
tis and some immunocompromising conditions. 13,34,35 Unlike contact with smallpox lesions should be destroyed or decon
ACAM2000, IMVAMUNE is administered subcutaneously in taminated. Smallpox virions are inactivated by exposure to
two doses separated by 4 weeks. 9,34 There are no data on its ultraviolet light and chemical disinfectants, such as bleach or
effectiveness in postexposure vaccination. While IMVAMUNE Lysol. 1
is likely to replace ACAM2000 and it has been added to the
US Strategic National Stockpile, the DoD has yet to formally
incorporate its use into standard military doctrine. 9,30 Conclusion
Despite its eradication in nature, smallpox remains a relevant
Once infection is established, targeted antiviral therapy is lim bioagent threat due to its high fatality rate, transmissibility,
ited, and management primarily consists of postexposure vac lack of immunity in the current population, development as
cination, isolation, and supportive care. Presently, there are a biological weapon by the former Soviet Union and likely
three antiviral therapies shown to have effectiveness against possession by other nearpeer states. Today’s medical pro
smallpox in animals and in vitro studies – but none have been viders are unlikely to have ever seen a case of smallpox and
tested in humans. Cidofovir and its recently FDAapproved therefore vigilant education and awareness of its clinical pre
9
prodrug brincidofovir (TEMBEXA) are viral DNA polymerase sentation, transmissibility, and management are paramount to
inhibitors shown to have in vitro activity against smallpox and any military biodefense plan. While not yet standard in the
have been effective in treating animal poxviruses. The main military, a newly approved nonreplicating smallpox vaccine
36
adverse effects include nausea, diarrhea, and abdominal pain, and smallpoxspecific antivirals are now available and should
Smallpox as a Bioagent | 127