Page 70 - JSOM Spring 2018
P. 70
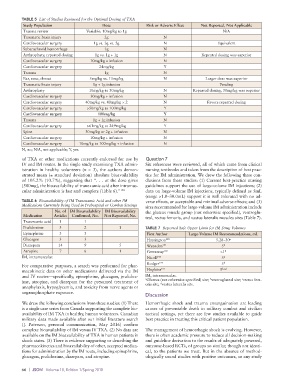
TABLE 5 List of Studies Reviewed for the Optimal Dosing of TXA
Study Population Dose Risk or Adverse Effect Not Reported, Not Applicable
Trauma review Variable: 10mg/kg to 1g N/A
Traumatic brain injury 2g N
Cardiovascular surgery 1g vs. 3g vs. 5g N Equivalent
Subarachnoid hemorrhage 1g N
Arthroplasty, repeated dosing 1g vs. 1g + 1g N Repeated dosing was superior
Cardiovascular surgery 10mg/kg + infusion N
Cardiovascular surgery 24mg/kg Y
Trauma 1g N
Ear, nose, throat 5mg/kg vs. 15mg/kg N Larger dose was superior
Traumatic brain injury 1g + 1g infusion Pending
Arthroplasty 10mg/kg to 30mg/kg N Repeated dosing, 30mg/kg was superior
Cardiovascular surgery 30mg/kg + infusion N
Cardiovascular surgery 40mg/kg vs. 40mg/kg × 2 N Favors repeated dosing
Cardiovascular surgery >50mg/kg to 100mg/kg Y
Cardiovascular surgery 100mg/kg Y
Trauma 1g + 1g infusion N
Cardiovascular surgery >61mg/kg to 249mg/kg Y
Spine 30mg/kg or 2g + infusion N
Cardiovascular surgery 10mg/kg + infusion N
Cardiovascular surgery 10mg/kg to 100mg/kg + infusion N
N, no; N/A, not applicable; Y, yes.
of TXA or other medications currently endorsed for use by Question 7
IV and IM routes. In the single study examining TXA admin- Six references were reviewed, all of which came from clinical
istration in healthy volunteers (n = 3), the authors demon- nursing textbooks and taken from the description of best prac-
strated mean (± standard deviation) absolute bioavailability tice for IM administration. We drew the following three con-
of 105.2% (10.7%), suggesting that “. . . at the dose given clusions from these studies: (1) Current best-practice nursing
[500mg], the bioavailability of tranexamic acid after intramus- guidelines support the use of large-volume IM injections; (2)
cular administration is fast and complete (Table 6).” 158 data on large-volume IM injections, typically defined as 5mL
(range >1.0–30.0mL) support it is well tolerated with no ad-
TABLE 6 Bioavailability of IM Tranexamic Acid and other IM verse effects, or acceptable and minimal adverse effects; and (3)
Medications Currently Being Used in Prehospital or Combat Settings sites recommended for large-volume IM administration include
No. of IM Bioavailability IM Bioavailability the gluteus muscle group (not otherwise specified), ventroglu-
Medication Articles Confirmed, No. Not Reported, No. teal, rectus femoris, and vastus lateralis muscles sites (Table 7).
Tranexamic acid 1 1
Pralidoxime 3 2 1 TABLE 7 Reported Safe Upper Limit for IM Drug Volumes
Epinephrine 3 3 First Author Large-Volume IM Recommendations, mL
Glucagon 3 3 Harrington 5.28–30 a
190
Diazepam 14 9 5 Wynaden 191 5 b
Atropine 7 6 1 Greenway 192 >1 b
IM, intramuscular. Nicoll 193 5 b
For comparative purposes, a search was performed for phar- Rodger 194 5 b
macokinetic data on other medications delivered via the IM Hopkins 195 5 b,c,d
and IV routes—specifically, epinephrine, glucagon, pralidox- IM, intramuscular.
Gluteus (not otherwise specified) site; ventrogluteal site; rectus fem-
ime, atropine, and diazepam for the presumed treatment of a oris site; vastus lateralis site. b c
d
anaphylaxis, hypoglycemia, and toxicity from nerve agents or
organophosphate exposure.
Discussion
We drew the following conclusions from these studies: (1) There Hemorrhagic shock and trauma exsanguination are leading
is a single case series from Canada supporting the complete bio- causes of preventable death in military combat and civilian
availability of IM TXA in healthy, human volunteers. Canadian tactical settings, yet there are few studies available to guide
military data made available after our initial literature search best practice in treating this critical patient population.
(J. Paterson, personal communication, May 2016) confirm
complete bioavailability of IM versus IV TXA. (2) No data are The management of hemorrhagic shock is evolving. However,
available on the IM bioavailability of TXA in human patients in there is often academic pressure to reduce all decision-making
shock states. (3) There is evidence supporting or describing the and guideline derivation to the results of adequately powered,
pharmacokinetics and bioavailability of other, accepted medica- outcome-based RCTs, of groups so similar, though not identi-
tions for administration by the IM route, including epinephrine, cal, to the patients we treat. But in the absence of method-
glucagon, pralidoxime, diazepam, and atropine. ologically sound studies with positive outcomes, or any study
66 | JSOM Volume 18, Edition 1/Spring 2018