Page 69 - JSOM Spring 2018
P. 69
hemorrhage and intracranial hemorrhage. 113, 126 One was the chart reviews (Table 4). One study ultimately was not included.
nested CRASH-2 intracranial bleeding study that did not dis- A broad range of TXA doses was reported, from 10mg/kg to
cuss the timing of TXA administration. One was the original 259mg/kg (Table 5). The most common nonweight-based regi-
129
CRASH-2 publication in Lancet. One looked at the timing men was 1g IV. Four observational studies reported potential
134
of TXA administration after arrival to hospital, however was adverse effects with TXA administration. All four studies were
unable to report the time of initial injury. The other excluded in patient cohorts undergoing major cardiovascular surgery
96
articles did not comment on the timing of TXA administration. (i.e., coronary artery bypass grafting, aortic valve replacement,
and/or cardiopulmonary bypass). Relatively large TXA doses
We drew two conclusions from these studies: (1) The only were used in each series (24mg/kg, >100mg/kg, 100mg/kg, and
prospective RCT data that compare timing of TXA admin- 61–259mg/kg), suggesting an increased risk for convulsive sei-
istration to patient outcomes were from the CRASH-2 study, zures in patients who underwent cardiovascular surgery.
suggesting that early treatment with TXA (within 3 hours of
injury [ideally, within 1 hour]) significantly reduced the risk of We drew the following conclusions from these studies: (1)
death due to bleeding; and (2) implementation of TXA pro- there is insufficient evidence to provide guidance on the mini-
tocols by military and civilian prehospital systems has been mal, maximal, or optimal dosing of TXA in major trauma or
described and is effective in administering TXA within 3 hours hemorrhagic shock; (2) TXA has been used in reported doses
of injury. of between 5mg/kg and 259mg/kg; (3) there is no evidence of
harm with multiple or repeat doses of TXA; (4) a few studies
All the included review articles, editorials, and perspective have demonstrated potential benefit of serial dosing of TXA
publications refer to the primary data from the Roberts et al. in operative settings; and (5) based on retrospective, observa-
exploratory analysis of the CRASH-2 trial. 132 tional case series, TXA used in patients undergoing cardiovas-
cular surgery who are placed on cardiopulmonary bypass and
Table 3 categorizes the articles as primary data, reviews, or receive >24mg/kg to 259mg/kg TXA may experience increased
opinions, in keeping with the CRASH-2 exploratory analysis more convulsive seizure.
and feasibility of timely prehospital TXA administration.
Question 6
Question 5 Thirty-one articles were obtained using the search strategy;
A total of 19 articles were obtained using the search strategy: of these, only one was specific to TXA. In general, there are
nine RCT or head-to-head trials, two review articles or opinion few strong studies demonstrating the relative pharmacokinet-
pieces, three prospective cohort studies, and five retrospective ics and pharmacodynamics after IV, IO, or IM administration
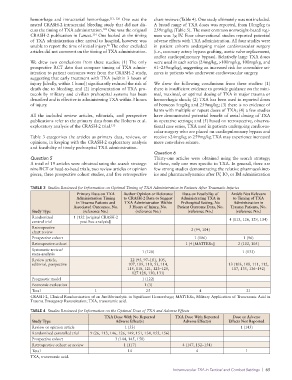
TABLE 3 Studies Reviewed for Information on Optimal Timing of TXA Administration in Patients After Traumatic Injuries
Primary Data on TXA Author Opinion or Reference Data on Feasibility of Article Not Relevant
Administration Timing to CRASH-2 Data to Suggest Administrating TXA in to Timing of TXA
in Trauma Patients and TXA Administration Within Prehospital Setting, No Administration in
Associated Outcomes, No. 3 Hours of Injury, No. Patient Outcome Data, No. Trauma Patients, No.
Study Type (reference No.) (reference No.) (reference No.) (reference No.)
Randomized 1 (132 [original CRASH-2 4 (113, 126, 129, 134)
control trial post hoc analysis])
Retrospective 2 (94, 104)
chart review
Prospective cohort 1 (106) 1 (96)
Retrospective cohort 1 (4 [MATTERs]) 2 (102, 105)
Systematic review/
meta-analysis 1 (120) 1 (133)
Review article, 22 (95, 97–101, 105,
editorial, perspective 107, 109, 110, 53, 114, 13 (103, 108, 111, 112,
115, 118, 121, 123–125, 117, 135, 136–142)
127 128, 130, 131)
Prognostic model 1 (122)
Economic evaluation 1 (3)
Total 1 25 4 21
CRASH-2, Clinical Randomization of an Antifibrinolytic in Significant Hemorrhage; MATTERs, Military Application of Tranexamic Acid in
Trauma Emergency Resuscitation; TXA, tranexamic acid.
TABLE 4 Studies Reviewed for Information on the Optimal Dose of TXA and Adverse Effects
TXA Dose With No Reported TXA Dose With Reported Dose or Adverse
Study Type Adverse Effect(s) Adverse Effect(s) Effects Not Reported
Review or opinion article 1 (53) 1 (143)
Randomized controlled trial 9 (26, 113, 146, 126, 149, 151, 134, 155, 156)
Prospective cohort 3 (144, 145, 150)
Retrospective cohort or review 1 (117) 4 (147, 152–154)
Total 14 4 1
TXA, tranexamic acid.
Intramuscular TXA in Tactical and Combat Settings | 65