Page 74 - JSOM Spring 2020
P. 74
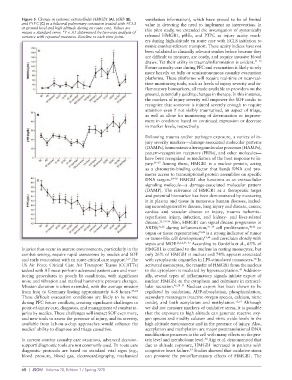
Figure 5 Change in systemic extracellular HMGB1 (A), pfHb (B), ventilation information), which have proved to be of limited
and PTPC (C) in a bilateral pulmonary contusion treated with ECLS value in detecting the need to implement an intervention. In
at ground level and high altitude during en route care. Values are this pilot study, we extended the investigation of systemically
means ± standard error. *P < .05 determined by two-way analysis of
variance with repeated measures. Baseline vs each time point. released HMGB1, pfHb, and PTPC as injury acuity mark-
ers during high-altitude en route care with ECLS initiation to
mimic combat-relevant transport. These acuity indices have not
been validated in clinically relevant studies before because they
are difficult to measure, are costly, and require invasive blood
draws. Yet their utility in trauma/inflammation is evident. 11–13
Future casualty care during PFC and evacuation is likely to rely
more heavily on fully or semiautonomous casualty evacuation
platforms. These platforms will require real-time or near-real-
time monitoring tools, such as levels of injury severity and in-
flammatory biomarkers, all made available to providers on the
ground, potentially guiding changes in therapy. In this instance,
the markers of injury severity will empower the SOF medic to
recognize that someone is injured severely enough to require
attention even if not visibly traumatized, an aspect of triage,
as well as allow for monitoring of deterioration or improve-
ment in condition based on continued expression or decrease
in marker levels, respectively.
Following trauma and/or pathogen exposure, a variety of in-
jury severity markers—damage-associated molecular patterns
(DAMPs), homeostasis-altering molecular processes (HAMPs),
pattern-recognition receptors (PRRs), and other molecules—
have been recognized as mediators of the host response to in-
jury. 25–27 Among these, HMGB1 is a nuclear protein, acting
as a chromatin-binding cofactor that bends DNA and pro-
motes access to transcriptional protein assemblies on specific
DNA targets. 28–30 HMGB1 also functions as an extracellular
signaling molecule—a damage-associated molecular pattern
(DAMP). The relevance of HMGB1 as a therapeutic target
and potential biomarker has been demonstrated by measuring
it in plasma and tissue in numerous human diseases, includ-
ing neurodegenerative disease, lung injury and disease, cancer,
cardiac and vascular disease or injury, trauma ischemia−
reperfusion injury, infection, and kidney- and liver-related
disease. 25,31–34 Also, HMGB1 can signal disease progression in
ARDS; 35,36 during inflammation, 37–39 cell proliferation, 40,41 or
organ or tissue regeneration; 39,42 is a strong indicator of tumor
or tumor-like cell development; 43,44 and correlates closely with
sepsis and MOF. 11,12,45–47 According to Gardella et al., 65% of
Injuries that occur in austere environments, particularly in the HMGB1 is confined to the nucleus in resting monocytes, but
combat setting, require rapid assessment by medics and SOF only 26% of HMGB1 is nuclear and 74% appears associated
48
and early evacuation with en route critical care support. The with cyto plasmic organelles in LPS-stimulated monocytes. In
1,2
US Air Force Critical Care Air Transport Teams (CCATTs) activated monocytes, the transfer of HMGB1 from the nucleus
49
tasked with AE must perform advanced patient care and mon- to the cytoplasm is mediated by hyperacetylation. Addition-
itoring procedures in poorly lit conditions, with significant ally, several types of inflammatory signals initiate export of
noise and vibration and marked barometric pressure changes. nuclear HMGB1 to the cytoplasm and culminate in extracel-
Mission duration is often extended, with the average mission lular secretion. 46,50–52 Nuclear export has been shown to be
from Iraq to Germany lasting approximately 6–8 hours. 23,24 regulated by oxidation, ADP-ribosylation, phosphorylation,
These difficult evacuation conditions are likely to be worse secondary messengers (reactive oxygen species, calcium, nitric
during PFC future conflicts, creating significant challenges in oxide), and both acetylation and methylation. 53,54 Although
point-of-injury care, diagnosis, and management of combat in- we did not measure markers of oxidative stress, it is possible
juries by medics. These challenges will impact SOF even more, that the exposure to high altitude can generate reactive oxy-
and new tools to assess the presence of injury, and its severity, gen species and modify calcium and nitric oxide levels in the
available from lab-on-a-chip approaches would enhance the high-altitude environment and in the presence of injury. Also,
medics’ ability to diagnose and triage casualties. acetylation and methylation are major posttranslational DNA
modification processes in the cell with many effects on the pro-
54
In current combat casualty care situations, advanced decision- tein level and metabolome level. Aigi et al. demonstrated that
support/ diagnostic tools are not commonly used. En route care due to altitude exposure, HMGB1 increased in patients with
55
diagnostic protocols are based on standard vital signs (e.g., congestive heart failure. Studies showed that oxidative stress
blood pressure, blood gas, electrocardiography, mechanical can promote the proinflammatory effects of HMGB1. The
68 | JSOM Volume 20, Edition 1 / Spring 2020