Page 56 - Journal of Special Operations Medicine - Fall 2014
P. 56
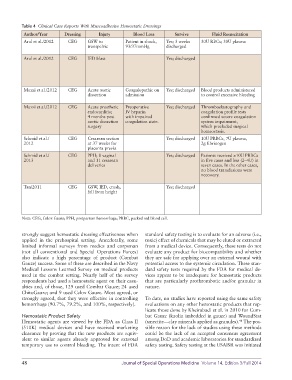
Table 4 Clinical Case Reports With Muscoadhesive Hemostatic Dressings
Author/Year Dressing Injury Blood Loss Survive Fluid Resuscitation Comments
Arul et al./2012 CEG GSW to Patient in shock, Yes; 3 weeks 30U RBCs; 30U plasma Case report 1: 25-year-old male; CEG used on combat casualties after conventional surgical attempts to achieve vascular
transpelvic 93/37mmHg discharged control in the pelvic region failed. Four rolls of CEG were packed into wound with direct pressure resulting in rapid
hemostasis.
Arul et al./2012 CEG IED blast Yes; discharged Case report 2: The Medical Emergency Response Team (MERT), Royal Air Force, Camp Bastion, Afghanistan. 22-year-old
male; two tourniquets were placed in the field; patient transferred to MERT; MERT personnel packed perineal region with
CEG due to severe bleeding. Patient arrived at field hospital in shock (BP 78/52mmHg; core temperature 32.9°C; blood pH
6.86). Bleeding could not be stopped with gauze wound packing and pelvic external fixation. Four rolls of CEG were then
packed into peritoneal cavity followed by standard gauze. Patient recovered weeks later. No complications reported with
prolonged CEG packing.
Muzzi et al./2012 CEG Acute aortic Coagulopathic on Yes; discharged Blood products administered Case report: 59-year-old male; despite the use of FFP, platelets, and pharmacological interventions, bleeding control was
dissection admission to control excessive bleeding not attained. CEG was cut into strips (10–20cm), which were used to pack the sternal edges and pericardium. After CEG
application, bleeding dramatically decreased. Coagulation parameters improved significantly over the next 36 hours.
Muzzi et al./2012 CEG Acute prosthetic Preoperative Yes; discharged Thromboelastography and Case report: 55-year-old male; same procedure with CEG strips used as described in preceding report. At 48 hours post
endocarditis; IV heparin coagulation profile tests surgery, bleeding stopped completely.
4 months post with impaired confirmed severe coagulation
aortic dissection coagulation state. system impairment, In this and the above case reports, the authors demonstrated the use of CEG to be lifesaving due to the compressive effect
surgery which precluded surgical with the ability to cause hemostasis in the presence of circulating heparin.
homeostasis.
Schmid et al./ CEG Cesarean section Yes; discharged 10U PRBCs, 7U plasma, Case report: 32-year-old female; 4 hours post surgery, vaginal bleeding continued after failed attempts at post 2 hours.
2012 at 37 weeks for 2g fibrinogen CEG was packed uterovaginal and left in place 36 hours; hemostasis achieved.
placenta previa
Schmid et al./ CEG PPH; 8 vaginal Yes; discharged Patients received ≥10U PRBCs Case reports: 19 cases of PPH due to uterine atony, placenta accreta/increta, or coagulopathy, including 5 severe cases where
2013 and 11 cesarean in five cases and less (2–4U) in a hysterectomy seemed inevitable otherwise. Celox Gauze left in place for 24–30 hours. In all but one case, the bleeding
deliveries seven cases. In the other cases, stopped and further interventions were avoided. Over comparable periods of time (18 months) and births (3822 vs. 4077)
no blood transfusions were before and after the introduction of the CEB in our clinic, the rate of peripartum hysterectomies was reduced by 75% (8 vs.
necessary. 2; odds ratio 4.27; p = .044). Celox Gauze is an effective option in the treatment of severe PPH. It is easy to use and requires
no special training. It can be used after both vaginal and cesarean deliveries, and there were no adverse side effects.
Tan/2011 CEG GSW, IED, crush, Yes; discharged Case reports: Dutch Field Hospital, Camp Holland, Afghanistan. Seven traumatic injuries are described with the application
fall from height of CEG during air medical evacuation and patients in the emergency department, or operating room; injuries were to lower
extremities, pelvic region, neck, ear and nose; in six of seven cases, CEG successfully stopped the bleeding. The fall from
a height caused head trauma, and CEG application was unsuccessful due to lack of vessel contact. Two patients had CEG
applied success to wounds for 12- and 24-hour durations without complications. Dutch medical personal preferred CEG for
ease of use and effectiveness; no side effects reported.
Note: CEG, Celox Gauze; PPH, postpartum hemorrhage; PRBC, packed red blood cell.
strongly suggest hemostatic dressing effectiveness when standard safety testing is to evaluate for an adverse (i.e.,
applied in the prehospital setting. Anecdotally, some toxic) effect of chemicals that may be eluted or extracted
limited informal surveys from medics and corpsman from a medical device. Consequently, these tests do not
(not all conventional and Special Operations Forces) evaluate any product for biocompatibility and whether
also indicate a high percentage of product (Combat they are safe for applying over an external wound with
Gauze) success. Some of these are described in the Navy potential access to the systemic circulation. These stan-
Medical Lessons Learned Survey on medical products dard safety tests required by the FDA for medical de-
used in the combat setting. Nearly half of the survey vices appear to be inadequate for hemostatic products
respondents had used a hemostatic agent on their casu- that are particularly prothrombotic and/or granular in
alties and, of those, 129 used Combat Gauze; 24 used nature.
ChitoGauze; and 9 used Celox Gauze. Most agreed, or
strongly agreed, that they were effective in controlling To date, no studies have reported using the same safety
hemorrhage (90.7%, 79.2%, and 100%, respectively). evaluations on any other hemostatic products that rep-
licate those done by Kheirabadi et al. in 2010 for Com-
Hemostatic Product Safety bat Gauze (kaolin imbedded in gauze) and WoundStat
Hemostatic agents are viewed by the FDA as Class II (smectite—clay minerals applied as granules). The pos-
48
(510K) medical devices and have received marketing sible reason for the lack of studies using these methods
clearance by proving that the new products are equiv- could be the lack of an accepted consensus agreement
alent to similar agents already approved for external among DoD and academic laboratories for standardized
temporary use to control bleeding. The intent of FDA safety testing. Safety testing at the USAISR was initiated
48 Journal of Special Operations Medicine Volume 14, Edition 3/Fall 2014