Page 84 - JSOM Summer 2025
P. 84
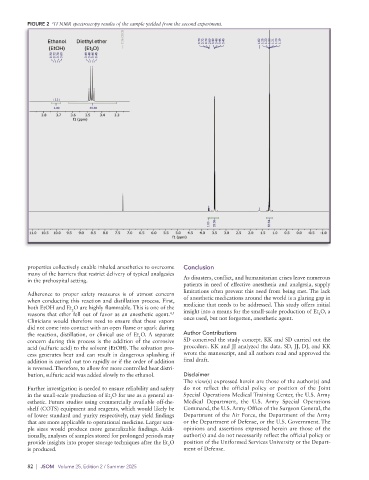
FIGURE 2 H NMR spectroscopy results of the sample yielded from the second experiment.
1
properties collectively enable inhaled anesthetics to overcome Conclusion
many of the barriers that restrict delivery of typical analgesics
in the prehospital setting. As disasters, conflict, and humanitarian crises leave numerous
patients in need of effective anesthesia and analgesia, supply
limitations often prevent this need from being met. The lack
Adherence to proper safety measures is of utmost concern
when conducting this reaction and distillation process. First, of anesthetic medications around the world is a glaring gap in
both EtOH and Et O are highly flammable. This is one of the medicine that needs to be addressed. This study offers initial
2
reasons that ether fell out of favor as an anesthetic agent. insight into a means for the small-scale production of Et O, a
4,5
2
Clinicians would therefore need to ensure that these vapors once used, but not forgotten, anesthetic agent.
did not come into contact with an open flame or spark during
the reaction, distillation, or clinical use of Et O. A separate Author Contributions
2
concern during this process is the addition of the corrosive SD conceived the study concept. KK and SD carried out the
acid (sulfuric acid) to the solvent (EtOH). The solvation pro- procedure. KK and JJ analyzed the data. SD, JJ, DJ, and KK
cess generates heat and can result in dangerous splashing if wrote the manuscript, and all authors read and approved the
addition is carried out too rapidly or if the order of addition final draft.
is reversed. Therefore, to allow for more controlled heat distri-
bution, sulfuric acid was added slowly to the ethanol. Disclaimer
The view(s) expressed herein are those of the author(s) and
Further investigation is needed to ensure reliability and safety do not reflect the official policy or position of the Joint
in the small-scale production of Et O for use as a general an- Special Operations Medical Training Center, the U.S. Army
2
esthetic. Future studies using commercially available off-the- Medical Department, the U.S. Army Special Operations
shelf (COTS) equipment and reagents, which would likely be Command, the U.S. Army Office of the Surgeon General, the
of lower standard and purity respectively, may yield findings Department of the Air Force, the Department of the Army
that are more applicable to operational medicine. Larger sam- or the Department of Defense, or the U.S. Government. The
ple sizes would produce more generalizable findings. Addi- opinions and assertions expressed herein are those of the
tionally, analyses of samples stored for prolonged periods may author(s) and do not necessarily reflect the official policy or
provide insights into proper storage techniques after the Et O position of the Uniformed Services University or the Depart-
2
is produced. ment of Defense.
82 | JSOM Volume 25, Edition 2 / Summer 2025