Page 119 - Journal of Special Operations Medicine - Fall 2015
P. 119
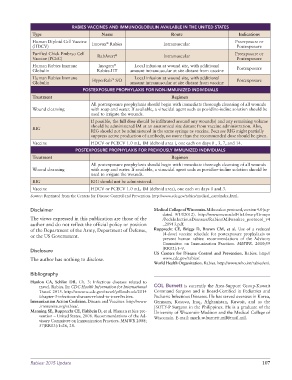
RABIES VACCINES AND IMMUNOGLOBULIN AVAILABLE IN THE UNITED STATES
Type Name Route Indications
Human Diploid Cell Vaccine Imovax Rabies Intramuscular Preexposure or
®
(HDCV) Postexposure
Purified Chick Embryo Cell RabAvert ® Intramuscular Preexposure or
Vaccine (PCEC) Postexposure
Human Rabies Immune Imogam Local infusion at wound site, with additional Postexposure
®
Globulin Rabies-HT amount intramuscular at site distant from vaccine
Human Rabies Immune HyperRab S/D Local infusion at wound site, with additional Postexposure
™
Globulin amount intramuscular at site distant from vaccine
POSTEXPOSURE PROPHYLAXIS FOR NON-IMMUNIZED INDIVIDUALS
Treatment Regimen
All postexposure prophylaxis should begin with immediate thorough cleansing of all wounds
Wound cleansing with soap and water. If available, a virucidal agent such as povidine-iodine solution should be
used to irrigate the wounds.
If possible, the full dose should be infiltrated around any wound(s) and any remaining volume
should be administered IM at an anatomical site distant from vaccine administration. Also,
RIG
RIG should not be administered in the same syringe as vaccine. Because RIG might partially
suppress active production of antibody, no more than the recommended dose should be given.
Vaccine HDCV or PCECV 1.0 mL, IM (deltoid area ), one each on days 0 , 3, 7, and 14.
POSTEXPOSURE PROPHYLAXIS FOR PREVIOUSLY IMMUNIZED INDIVIDUALS
Treatment Regimen
All postexposure prophylaxis should begin with immediate thorough cleansing of all wounds
Wound cleansing with soap and water. If available, a virucidal agent such as povidine-iodine solution should be
used to irrigate the wounds.
RIG RIG should not be administered.
Vaccine HDCV or PCECV 1.0 mL, IM (deltoid area), one each on days 0 and 3.
Source: Reprinted from the Centers for Disease Control and Prevention. http://www.cdc.gov/rabies/medical_care/index.html.
Disclaimer Medical College of Wisconsin. Milwaukee protocol, version 4.0 (up-
dated 9/13/2012). http://www.mcw.edu/FileLibrary/Groups
The views expressed in this publication are those of the /PedsInfectiousDiseases/Rabies/Milwaukee_protocol_v4
author and do not reflect the official policy or position _20913.pdf.
of the Department of the Army, Department of Defense, Rupprecht CE, Briggs D, Brown CM, et al. Use of a reduced
or the US Government. (4-dose) vaccine schedule for postexposure prophylaxis to
prevent human rabies: recommendations of the Advisory
Committee on Immunization Practices. MMWR. 2010;59
Disclosure (RR02):1–9.
US Centers for Disease Control and Prevention. Rabies. http://
The author has nothing to disclose. www.cdc.gov/rabies/.
World Health Organization. Rabies. http://www.who.int/rabies/en/.
Bibliography
Hanlon CA, Schlim DR. Ch. 3: Infectious diseases related to
travel. Rabies. In: CDC Health Information for International COL Burnett is currently the Area-Support Group-Kuwait
Travel. 2015. http://wwwnc.cdc.gov/travel/yellowbook/2014 Command Surgeon and is Board-Certified in Pediatrics and
/chapter-3-infectious-diseases-related-to-travel/rabies. Pediatric Infectious Diseases. He has served overseas in Korea,
Immunization Action Coalition. Disease and Vaccines. http://www Germany, Kosovo, Iraq, Afghanistan, Kuwait, and as the
.immunize.org/rabies/. JSOTF-P Surgeon in the Philippines. He is a graduate of the
Manning SE, Rupprecht CE, Fishbein D, et al. Human rabies pre- University of Wisconsin-Madison and the Medical College of
vention – United States, 2008. Recommendations of the Ad- Wisconsin. E-mail: mark.w.burnett.mil@mail.mil.
visory Committee on Immunization Practices. MMWR 2008;
57(RR03):1–26, 28.
Rabies: 2015 Update 107