Page 359 - ATP-P 11th Ed
P. 359
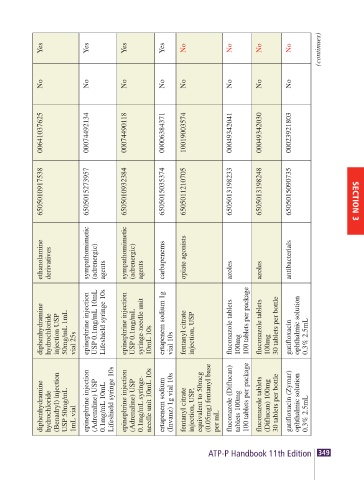
JDF Status No Yes Yes Yes Yes Yes Yes Yes Yes Yes Yes No No No No (continues)
Controlled No Yes Yes Yes Yes No No No No No No No No No No
Recommended NDC 08290328230 51079028521 00409321302 00409127332 00555005902 00409229031 00641037625 00074492134 00074490118 00006384371 10019003574 00049342041 00049342030 00023921803
6505014253165 SECTION 3
NSN 6505010985802 6505012740951 6505015138434 6505015053476 6505001168350 6505015182962 6505010917538 6505015273957 6505010932384 6505015035374 6505011210705 6505013198233 6505013198248 6505015090735
AHFS Category caloric agents benzodiazepines benzodiazepines benzodiazepines benzodiazepines ethanolamine derivatives ethanolamine derivatives ethanolamine derivatives sympathomimetic (adrenergic) agents sympathomimetic (adrenergic) agents carbapenems opiate agonists azoles azoles anti
MASTER DRUG LIST (cont.) Nomenclature Common Name dextrose tablets 45g dextrose tablets 45g multi-use squeze tube multi-use squeeze tube 12 tablets diazepam 5mg tablets diazepam (Valium) I.S. 100s 5mg tablets I.S. 100s diazepam 5mg/mL, diazepam (Valium) 2mL autoinjector 5mg/mL, 2mL (cana) autoin
carpuject 10s
2mL syringe
0.3% 2.5mL
12 tablets
MDV 5s
per mL
1mL vial ATP-P Handbook 11th Edition 349